|
Specific Gravity of Solids
The density of a substance is its mass per unit volume,
i.e., its
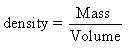
The Specific Gravity of a substance is the ratio of the
weight of nay volume of the substance to the weight of an equal
volume of water at 4°C.
Specific gravity of a substance
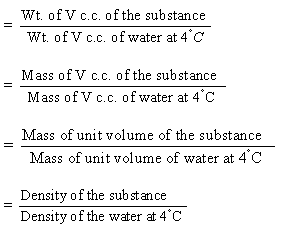
The
specific gravity of a substance is really a relative density, i.e.
its relative density to that of water at 4°C
In
C.G.S. system, the mass of 1 c.c. of water at 4°C is one gram, its
density is, therefore, 1 gm. per c.c.
In
M.K.S. it is 1 kg. per liter
In
F.P.S. system, the mass of 1 c.ft. of water is 62.5 lbs. or the
density is 62.5 lb. per c.ft.
Hence
in C.G.S. system, the density of a substance is numerically equal to
its specific gravity. On the other hand, the density of a substance
in F.P.S. unit is numerically equal to 62.5 X specific gravity of the
substance.
back to Top
Determination
of Specific Gravity
a)
By Archimedes' Principle:
1)
To find the Sp. Gravity of solid heavier than water.
Let
the weight of the solid in air = W1 gm.
Weight
of the solid in water= W2 gm
The
weight of the same volume of water as that of the solid = (W1-W2)
gms.
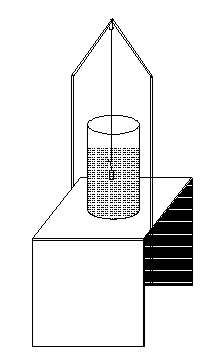
To
take the weight of the body in water, it is suspended by means of a
fine thread from the hook of the left pan and made to sink completely
in water contained in a beaker. The beaker is placed on a small
wooden bridge, which is put across the pan in such a way that the
bridge or the beaker does not touch any part of the pan of the
balance.
Therefore,
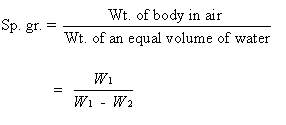
2)
Solid Lighter than Water
Let
the weight of solid in air = W1 gm
Take
another heavy body, called a sinker, such that the two tied together
may sink in water.
Let
the weight of the solid and sinker both in water = W2 gm
Weight
of the sinker of the solid and sinker both in water = W3
gm
Weight
of solid in water = W2 - W3 gms.
=
W1 × (W2 W3) = W1 × (W2
W3)
Therefore,
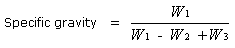
3) Solid Soluble in Water
The specific gravity of a solid soluble in water can be found by
immersing the solid in a liquid of known specific gravity in which the
solid is insoluble.
Determine the specific
gravity of the solid relative to the liquid. Then the actual specific
gravity of the solid will be obtained by multiplying this value with
the specific gravity of the liquid.
Specific gravity of solid

where
W1 = wt. of solid in air;
W2 = wt. of solid in the given liquid:
d = sp. gr. of the liquid.
back to Top
b)
By the Specific Gravity Bottle.
It is a small
glass bottle usually of 25 or 50 c.c. capacity with a slight conical
neck. It is fitted with a ground glass stopper which has a fine bore
along the whole length. When the bottle is completely filled with any
liquid and the stopper is fitted to the mouth, the excess of the
liquid flows out through the hole in the stopper. Thus, the inside
volume of the bottle is always kept constant. This bottle is used to
find out the specific gravity of a solid in the form of powder or
small fragments, and of liquid also.
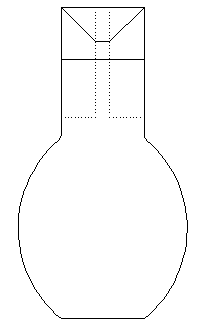
Let the
weight of the empty bottle = W1 gm.
The
weight of the bottle +powder put inside = W2
gm.
Weight
of the powder = (W2-W1) gm.
Weight
of the bottle + powder water to fill the rest of the
bottle = W3 gm.
Now
pour out all the contents of the bottle and fill it up
with pure water taking care to remove any air bubbles from
inside. Let the weight of the bottle when full of water =W4
gm.
Weight
of an equal volume of water as that of the powder = (W4-W1)
(W3-W2) gm.
Hence Sp. Gr. =
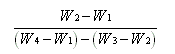
back to Top
|